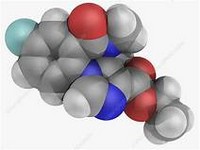
Fluvastatin

Fluvastatin
CLINICAL USE
HMG CoA reductase inhibitor:Primary hypercholesterolaemia Slowing progression of atherosclerosis Secondary prevention of coronary events after percutaneous coronary interventionDOSE IN NORMAL RENAL FUNCTION
20–80 mg daily in the eveningXL: 80 mg dailyPHARMACOKINETICS
DOSE IN RENAL IMPAIRMENT
GFR (mL/MIN)
DOSE IN PATIENTS UNDERGOING RENAL REPLACEMENT THERAPIES
IMPORTANT DRUG INTERACTIONS
Potentially hazardous interactions with other drugsADMINISTRATION
Reconstition
–Route
OralRate of Administration
–Comments
–OTHER INFORMATION
The Committee on Safety of Medicines has advised that rhabdomyolysis associated with lipid-lowering drugs, such as the fibrates and statins, appears to be rare (approx. 1 case in every 100 000 treatment years), but may be increased in those with renal impairment and possibly in those with hypothyroidismManufacturer’s literature indicates fluvastatin is contraindicated in patients with severe renal impairment (creatinine greater than or equal to 160 µmol/L).
See how to identify renal failure stages according to GFR calculation
See how to diagnose irreversible renal disease
Home